 | |
 | |
| Combination of | |
|---|---|
| Oxycodone | Opioid analgesic |
| Naloxone | Opioid receptor antagonist |
| Clinical data | |
| Trade names | Targin, Targiniq, Targinact, others |
| AHFS/Drugs.com | International Drug Names UK Drug Information |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| (verify) | |
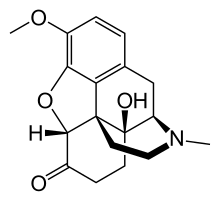
Oxycodone/naloxone, sold under the brand name Targin among others, is a combination pain medication. It is available as modified-release tablets and is taken by mouth.
The oxycodone component is an opioid and is responsible for the pain-relieving effects. Naloxone opposes the effects of opioids but is poorly absorbed into the body when given orally, meaning almost all the dose stays within the gastrointestinal tract and reduces the local side effects from the oxycodone, namely constipation as the naloxone binds to the opioid receptors in the gut, preventing the opioid from attaching. This does not affect its analgesic efficacy compared to Oxycontin. Constipation was significantly relieved in a 2008 study. The drug was released in 2006 in Germany and is available in some other European countries since 2009. In the UK, the 10 mg oxycodone / 5 mg naloxone and 20 mg/10 mg strengths were approved in December 2008, and the 40 mg/20 mg and 5 mg/10 mg strengths in July 2019.
Preliminary evidence suggests that oxycodone/naloxone may be an effective treatment for severe, refractory restless legs syndrome if first-line therapies have not been effective.
Adverse effects
References
- ^ "Public Summary". www.ebs.tga.gov.au. Australian Government, Department of Health and Aged Care. Retrieved 2024-05-20.
- ^ "Oxonal (AU Pharma Pty Ltd)". Therapeutic Goods Administration (TGA). 28 September 2022. Archived from the original on 18 March 2023. Retrieved 9 April 2023.
- ^ "Targin Product information". Health Canada. 22 October 2009. Archived from the original on 29 April 2023. Retrieved 9 April 2023.
- ^ "Targinact 5 mg/2.5 mg prolonged-release tablets - Summary of Product Characteristics (SmPC)". (emc). 16 March 2021. Archived from the original on 10 April 2023. Retrieved 9 April 2023.
- ^ a b "Targiniq ER- oxycodone hydrochloride/naloxone hydrochloride tablet, film coated, extended release". DailyMed. 22 December 2016. Archived from the original on 18 March 2023. Retrieved 18 March 2023.
- ^ Simpson K, Leyendecker P, Hopp M, Müller-Lissner S, Löwenstein O, De Andrés J, et al. (December 2008). "Fixed-ratio combination oxycodone/naloxone compared with oxycodone alone for the relief of opioid-induced constipation in moderate-to-severe noncancer pain". Current Medical Research and Opinion. 24 (12): 3503–3512. doi:10.1185/03007990802584454. PMID 19032132. S2CID 73061000. Archived from the original on 2013-01-26. Retrieved 2009-04-09.
- ^ Mundipharma (2009-01-26). "Targin (oral oxycodone/naloxone prolonged-release tablet) now launching across Europe to control severe chronic pain with significantly reduced risk of opioid-induced constipation". Archived from the original on 2009-03-17. Retrieved 2009-04-09.
- ^ de Biase S, Valente M, Gigli GL (2016). "Intractable restless legs syndrome: role of prolonged-release oxycodone-naloxone". Neuropsychiatric Disease and Treatment. 12: 417–425. doi:10.2147/NDT.S81186. PMC 4770072. PMID 26966363.
- ^ Trenkwalder C, Beneš H, Grote L, García-Borreguero D, Högl B, Hopp M, et al. (December 2013). "Prolonged release oxycodone-naloxone for treatment of severe restless legs syndrome after failure of previous treatment: a double-blind, randomised, placebo-controlled trial with an open-label extension". The Lancet. Neurology. 12 (12): 1141–1150. doi:10.1016/S1474-4422(13)70239-4. PMID 24140442. S2CID 35122538.
- ^ de Oliveira CO, Carvalho LB, Carlos K, Conti C, de Oliveira MM, Prado LB, Prado GF (June 2016). "Opioids for restless legs syndrome". The Cochrane Database of Systematic Reviews. 2016 (6): CD006941. doi:10.1002/14651858.CD006941.pub2. PMC 6885031. PMID 27355187.